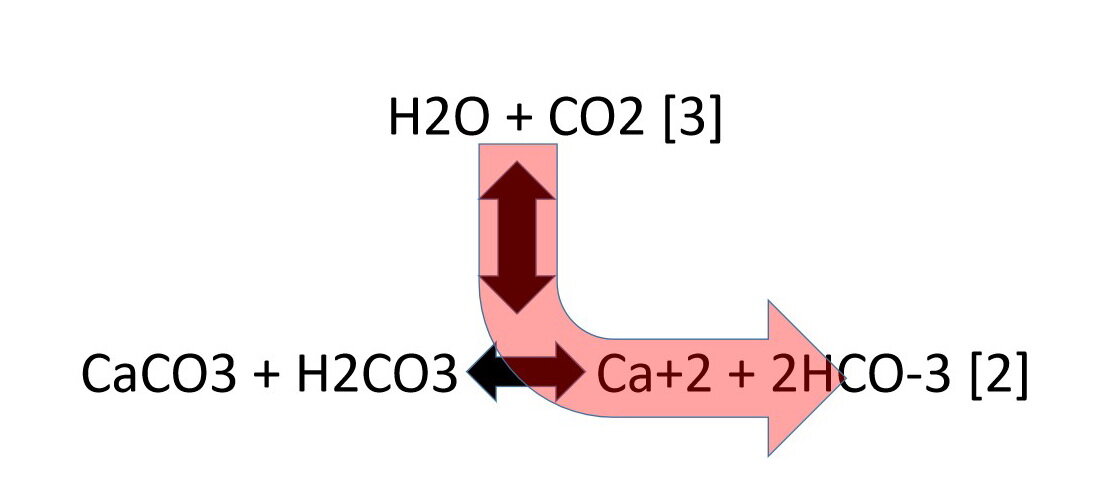
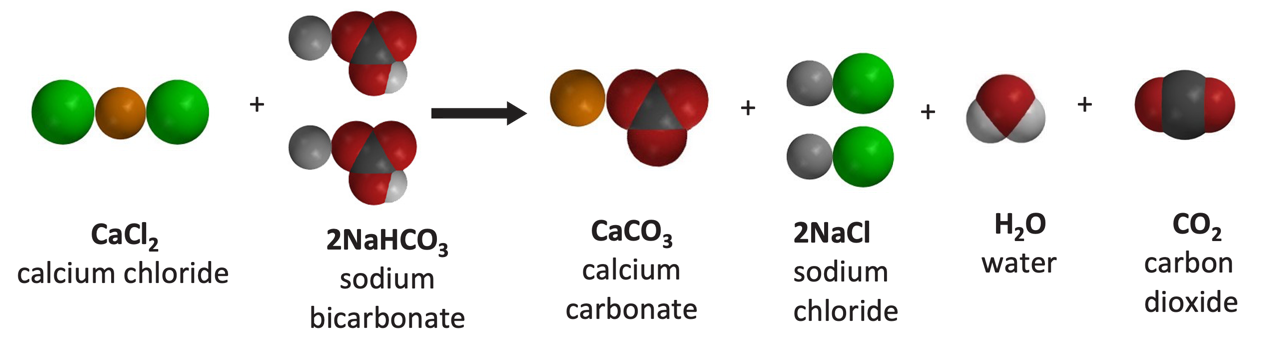
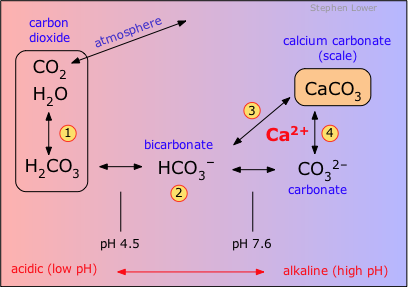
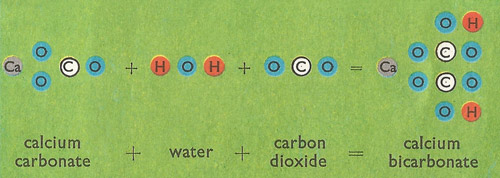
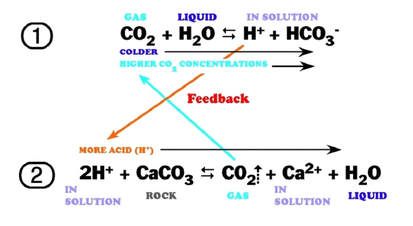
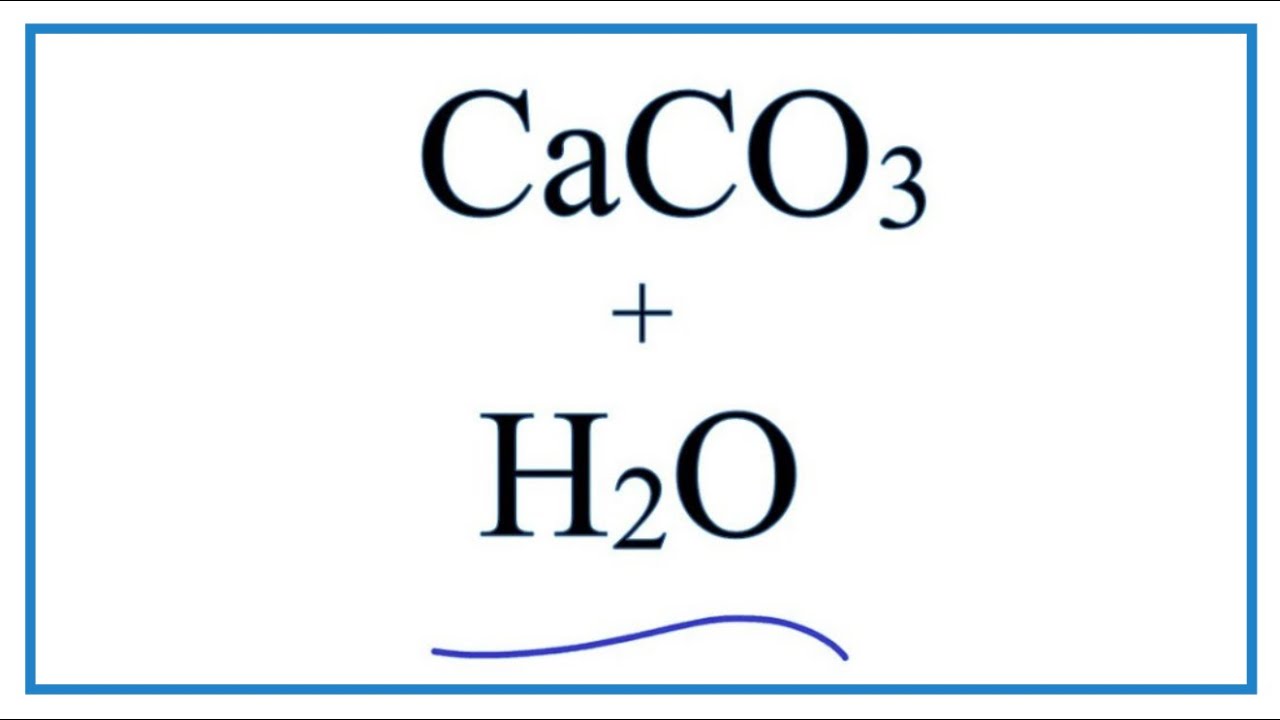
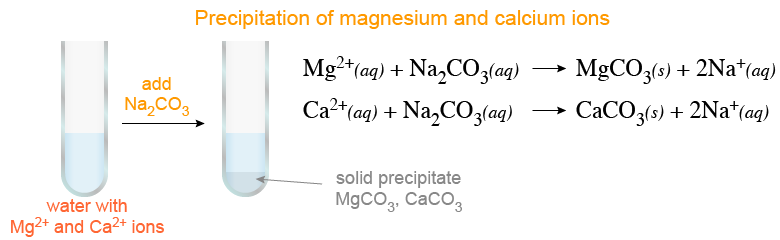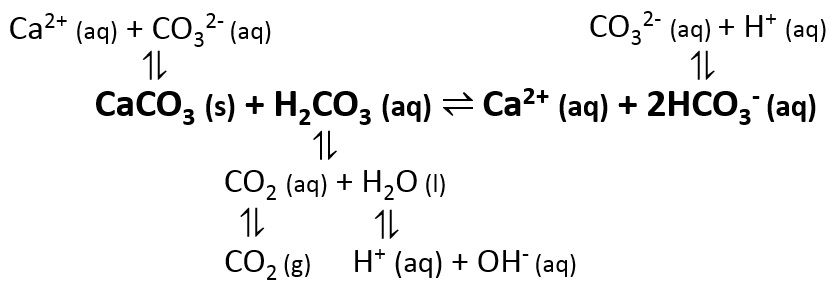Calcium carbonate reacts with hydrochloric acid to produce calcium chloride, water, and carbon dioxide gas. What is the balanced equation for it? - Quora

Writing a Net Ionic Equation for the Reaction of Solid Calcium Carbonate with a Hydrochloric Acid Solution

Schematic of formation of quick lime, hydrate lime, and calcium carbonate | Download Scientific Diagram
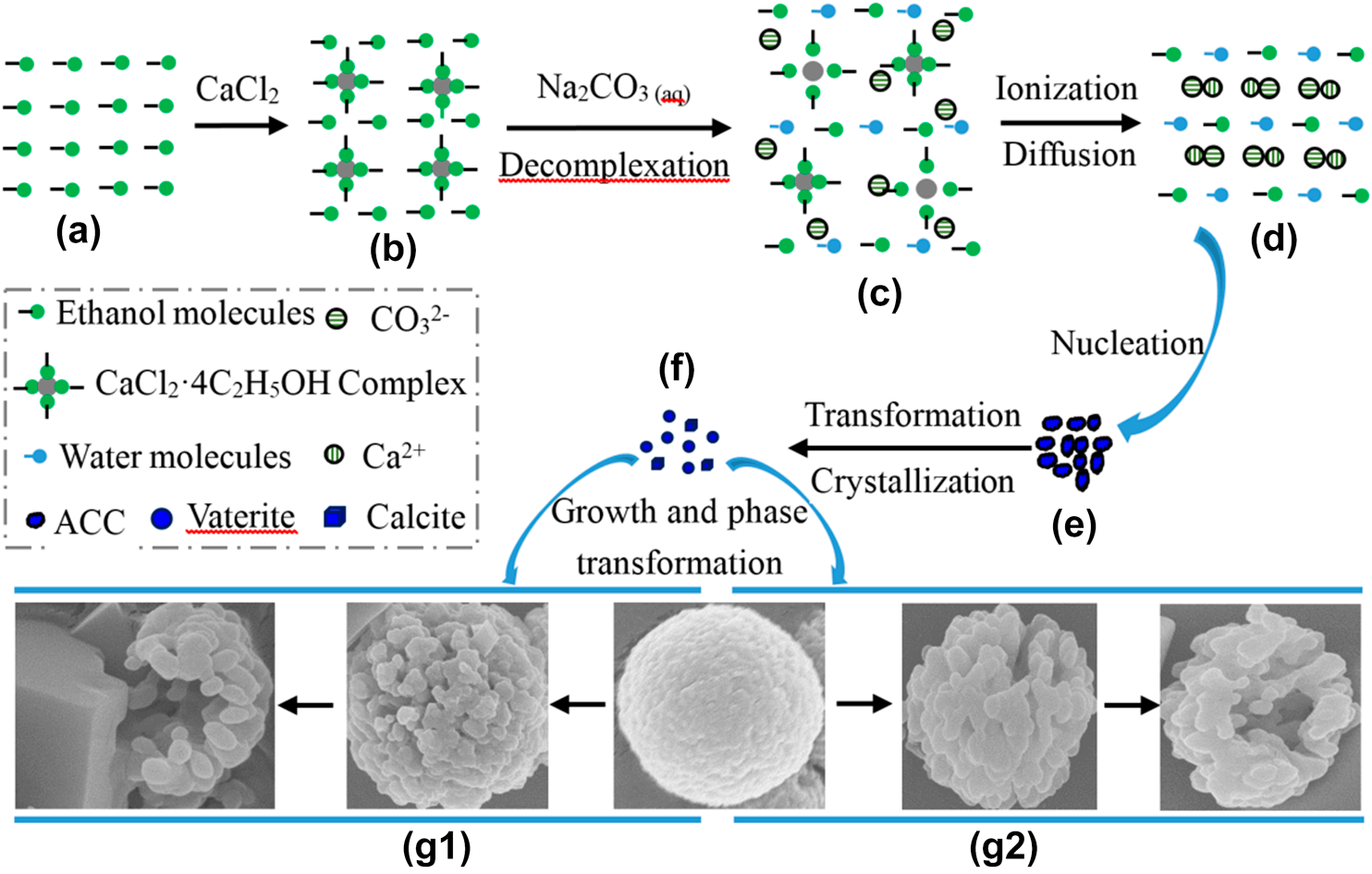
The advantage of alcohol–calcium method on the formation and the stability of vaterite against ethanol–water binary solvent method | Journal of Materials Research | Cambridge Core

One method of determining the proportion of calcium carbonate in a coral is to dissolve a known mass of the coral in excess acid and measure the volume of carbon dioxide formed.